Legal Notice Regarding This Product
THIS PRODUCT IS STRICTLY INTENDED FOR RESEARCH USE ONLY. It is designated solely as a research chemical. Usage is confined to in vitro testing and laboratory experimentation exclusively. Information available on our website about this product is for educational purposes only and should not be interpreted as an endorsement or directive for use outside these bounds. Introducing this product into humans or animals by any route is strictly prohibited by law. Handling of this product should be undertaken only by licensed, qualified professionals.
This product is not to be used as a drug, food, or cosmetic. Misbranding, misusing, or mislabeling this product as a drug, food, or cosmetic is illegal. Users are responsible for complying with all applicable laws and regulations regarding the handling and use of this product.
THE PRODUCTS PURCHASED ON THIS WEBSITE ARE INTENDED FOR RESEARCH CHEMICAL USE ONLY. These products are not intended to be used for human or animal consumption and/or ingestion of any kind. These products should not be used as food additives, drugs or household chemicals. These products should only be used by Qualified Professionals.
Bodily introduction into Human and Animals of any kind is strictly forbidden by law.
All the product information on this website is for educational purpose only.
Product Usage: THE PRODUCT IS INTENDED AS A RESEARCH PURPOSE ONLY. This designation allows the use of research chemical strictly for in vitro testing and laboratory experimentation only. All product information available on www.peptidesworld.com is for educational purposes only. Bodily introduction of any food, cosmetic and may not be misbranded, misused or mislabeled as a drug, food or cosmetics.
Highlights:
- Humanin and Lifespan extension
- Coronary heart disease and heart failure·
- Arteriosclerosis
- Alzheimer
- Cancer and kidney disease
A new study led by researchers at the USC Leonard Davis School of Gerontology is the first to demonstrate that a tiny protein has a big impact on health and longevity in both animals and humans. [1]
The researchers examined humanin, a peptide encoded in the small genome of mitochondria — the powerhouses of the cell. From experiments in laboratory animals to measurements in human patients, the multi-site collaboration demonstrates how higher levels of humanin in the body are connected to longer lifespans and better health. It is linked to a lower risk for diseases such as Alzheimer’s.
“Humanin has long been known to help prevent many age-related diseases, and this is the first time that it has been shown that it can also increase lifespan,” said senior author Pinchas Cohen, professor of gerontology, medicine and biological sciences and dean of the USC Leonard Davis School.
An intriguing evolutionary history
Humanin has been found not only in human mitochondria but also throughout the animal kingdom, a sign that its related gene has been maintained, or conserved, throughout evolution. The study, which was published online in the journal Aging on June 23, examined humanin in several animal species, including worms and mice, as well as humans, including Alzheimer’s patients and children of centenarians.
The results highlight the potential for humanin and other mitochondrial proteins to become treatments for age-related ailments. They also indicate that humanin may be an ancient mitochondrial signaling mechanism that is key for regulating the body’s health and lifespan, said first author and USC Leonard Davis Research Assistant Professor Kelvin Yen.
More humanin, longer lifespans
Humanin levels have previously been observed to decrease with age in many species. In this new study, the scientists observed higher levels of humanin in organisms predisposed to long lives, including the famously age-resistant naked mole rat, which experiences only a very slow decline in levels of humanin circulating in the body throughout its 30-year lifespan.
In contrast, mice experience a 40% drop in humanin over the first 18 months of life, and primates such as rhesus macaques appeared to have a similarly dramatic drop in humanin between the ages of 19 and 25.
In humans, researchers observed this phenomenon of higher and more sustained levels of humanin in 18 children of centenarians, versus a control group of 19 children of non-centenarians. Individuals whose parents reach 100 years old are statistically more likely than other people to reach very old age.
In some species, including worms and mice, modifying their genes to produce higher amounts of humanin within their bodies was enough to significantly increase lifespans. But these longer-lived animals had fewer offspring. Scientists have observed a similar pattern in long-lived humans.
“This tradeoff between longevity and reproduction is thought to be due to an evolutionarily conserved balance between using energy to produce more offspring or using the energy to maintain the organism for future reproductive efforts,” Yen said. “Evolutionarily speaking, the goal of life is to reproduce and then you’re done, but if you can’t reproduce, you should try to hang around as long as possible, and a side effect of that is longevity.”
Indication of — and protection against – Alzheimer disease
Higher humanin levels aren’t just linked to increased lifespan; lower levels may increase the risk of disease and lower resistance to toxic exposures.
The researchers analyzed samples of cerebral spinal fluid from a small number of Alzheimer’s patients and control individuals without dementia and noticed that humanin levels were much lower in the Alzheimer’s patients. And in newborn cord blood samples, high levels of humanin correlated with a high mitochondrial DNA (mtDNA) copy number, or the number of copies of the mitochondrial genome present within each cell.
Bone Strength
Humanin treatment, a potential new strategy to prevent bone growth impairment in chronic inflammatory disorders.
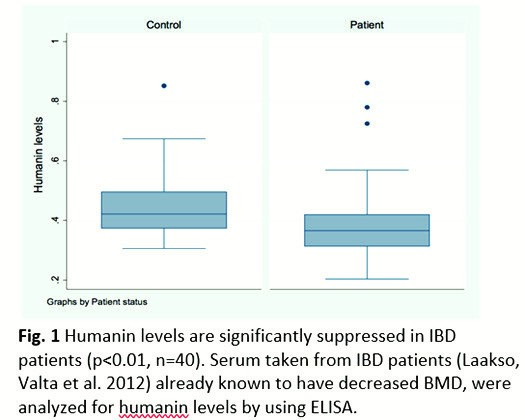
The data suggests that systemic levels of humanin are decreased in patients with chronic inflammation who suffer from bone growth impairment. Interestingly, the human analogue HNG was found to partially prevent cytokine-induced growth impairment in ex vivo cultured rat metatarsal bones. Our findings suggest that humanin is a potential drug target for the prevention of bone growth impairment in conditions of chronic inflammation. [3]
Cancer, Kidney disease and Cardiovascular disease
“Humanin levels are inversely correlated with a decrease in mitochondrial DNA (mtDNA) copy number, which in itself has been associated with a number of different diseases such as cancer, kidney disease, and cardiovascular disease,” Yen said.
Potential for treatments
Cohen’s laboratory was one of three groups that independently discovered humanin and has continued to unlock the secrets of the mitochondrial genome. Other promising mitochondrial peptides characterized by Cohen’s team include MOTS-c, which plays a role in communication between the mitochondria and the nucleus in cells and appears to mimic the effects of exercise.
This new wide-ranging study highlights the importance of humanin as a potentially powerful regulator of lifespan and health, and harnessing it for treatments could address a variety of age-related illnesses, Cohen said.
“This study, as well as many others, suggest that humanin administration would be an effective therapeutic treatment for a large number of diseases and further solidifies the importance of the mitochondria beyond its traditional role as the ‘powerhouse of the cell,'” he said.
Coronary Heart Disease and Heart Failure [2]
Oxidative stress is also involved in the pathogenesis of acute myocardial infarction and ischemia–reperfusion injury. HN protects cardiomyocytes from apoptosis through the antioxidation pathway, reducing the myocardial infarction size and improving cardiac function . HN also reduces the necrosis area of myocardial infarction and improves the cardiac function after myocardial infarction by reducing ROS production, which protects the function of myocardial mitochondria . HN has been shown to protect isolated myocardial mitochondria from H2O2-induced oxidative stress. HN increased the levels of GSH, GPX, and SOD, reversing myocardial ischemia–reperfusion injury . HNG upregulated the Akt/glycogen synthase kinase-3β pathway and inhibited myocardial fibrosis in aged mice . It has been found that Nrf2 and Keap1 are necessary for increasing the expression of SOD, CAT, GPX, and GSH. HN may promote the activation of Nrf2 by inhibiting the expression of Keap1 during myocardial infarction . In addition, the population study found that compared with normal people, the level of humanin in patients with coronary heart disease decreased and the level of lactic acid increased, suggesting that the protective effect of humanin on cardiovascular system is through antioxidant effect . Humanin is positively correlated with coronary artery endothelial function, which may be a target for the treatment of coronary heart disease in the future. [2]
Heart failure is the most common complication of myocardial infarction. HN has been shown to decrease the incidence rate of heart failure by inhibiting myocardial hypertrophy . Endonuclease G deficiency induces cardiomyocyte hypertrophy by increasing ROS production. Intriguingly, HN has been demonstrated to inhibit cardiomyocyte hypertrophy induced by endonuclease G deficiency [2]
Arteriosclerosis
Atherosclerosis is an age-related disease. HN and its potent analogs have beneficial effects against age-related diseases. Endothelial dysfunction contributes to atherosclerosis. Importantly, HN improves endothelial dysfunction through antioxidation, because (1) HN inhibits NOX2, thereby decreasing mitochondrial ROS production; (2) NLRP3 inflammasome activated by mitochondrial ROS leads to endothelial injury; however, HN inhibits the activation of NLRP3 inflammasome by activating AMPK. [2]
Hypercholesterolemia is involved in atherosclerosis because ox-LDL infiltrates the subendothelium to form atherosclerotic plaques after endothelial cell injury. However, HN prevents the progression of atherosclerotic plaques in hypercholesterolemic mice with apolipoprotein E (APOE) deficiency by reducing the level of nitrotyrosine (NT) and increasing the expression of endothelial nitric oxide synthase (eNOS), which are involved in oxidative stress. Ox-LDL is formed by ROS-related oxidation of LDL, ultimately promoting the formation and progression of atherosclerotic plaques by increasing lipid and cholesterol accumulation. Ox-LDL increases the expression of p62 and LC3-II and inhibits the function of cathepsin D activity. HN inhibits the ox-LDL-induced lipid and cholesterol accumulation by decreasing the LC3-II and p62 levels and restoring the ox-LDL-induced cathepsin D functional impairment, thereby reducing the formation of atherosclerotic plaques. Lectin-like oxidized low-density lipoprotein-1 (LOX-1) is the main receptor involved in absorption of ox-LDL by endothelial cells. LOX-1 mediates the binding, internalization, and proteolytic degradation of ox-LDL by endothelial cells. The HNG-induced decrease in LOX-1 protein expression also contributes to the inhibition of the formation and progression of atherosclerotic plaques. [2]
High glucose levels are also implicated in atherosclerosis, leading to endothelial dysfunction. High glucose increases ROS production and the expression of pro-inflammatory factors (tumor necrosis factor-α and IL-1β), further promoting endothelial cells to produce vascular cell adhesion molecule-1 (VCAM-1) and E-selectin. VCAM-1 and E-selectin mediate the adhesion of circulating leukocytes to the endothelium, leading to atherosclerosis development. Kruppel-like factor 2 (KLF2) is involved in endothelial dysfunction induced by high glucose. HN upregulates the KLF2 gene expression, inhibiting monocyte adhesion to endothelial cells. [2]
Journal Reference:
- Kelvin Yen, Hemal H. Mehta, Su-Jeong Kim, YanHe Lue, James Hoang, Noel Guerrero, Jenna Port, Qiuli Bi, Gerardo Navarrete, Sebastian Brandhorst, Kaitlyn Noel Lewis, Junxiang Wan, Ronald Swerdloff, Julie A. Mattison, Rochelle Buffenstein, Carrie V. Breton, Christina Wang, Valter Longo, Gil Atzmon, Douglas Wallace, Nir Barzilai, Pinchas Cohen.The mitochondrial derived peptide humanin is a regulator of lifespan and healthspan. Aging, 2020; DOI: 18632/aging.103534
- He Cai, Hongbo Men and Yang Zheng,
Protective Mechanism of Humanin Against Oxidative Stress in Aging-Related Cardiovascular Diseases
Front. Endocrinol., 10 June 2021 |DOI: https://doi.org/10.3389/fendo.2021.683151
- Zhao
Humanin treatment, a potential new strategy to prevent bone growth impairment in chronic inflammatory disorders, Journal of Orthopaedics Trauma Surgery and Related

Humanin 10mg
$119.90